09.04.2021by Matteo Pasini
Spin, meet Photon. Photon, meet Spin

Making light interact with matter at the quantum level with cavities and colour centres.
by Matteo Pasini
Many phenomena that we take for granted in the macroscopic world, like sun rays shining on dust particles suspended in air, or our image reflected in a mirror, become more complicated when we move to the quantum world. When instead of an intense and diffuse light beam we have a single photon, and when we want to shine it not anymore on a dust particle but on an Angstrom-sized object, things are not so straightforward. However, enhancing and exploiting the interaction between atomic-like quantum systems and single photons is a crucial task for quantum technologies: it allows to connect platforms that realise different quantum tasks, which enables long distance transmission of quantum information and, ultimately, makes a quantum internet possible.
(Don’t know yet what a quantum internet is? Read this: [1])
Qubits – the quantum counterpart of a single unit of classical information, the bit – can be arranged into two functional categories: stationary and flying qubits [2]. The name already give a hint of the properties we expect from each family. A good stationary qubit is, generally, one that is easy to manipulate, reliable and has a long memory. Stationary qubits are meant to be a platform for realising quantum operations, or a system where we can store a quantum state for a reasonably long time. From flying qubits, instead, we want mainly one thing: they need to fly. This is the tool we want to use for transmitting quantum information between distant points, hence we want it to be mobile, robust, insensitive to the external environment and, of course, fast.
There’s plenty of promising candidates for the role of “best stationary qubit”. They’re usually based on material platforms like atoms, impurities in crystals, superconducting circuits. In the race for the best flying qubit, by contrast, there’s only one contender. And surprisingly (or not) it’s the same that serves as pillar of classical information transmission: light.
The backbone of the classical internet (the one you are using to read this article) is based on pulses of light that travel through optical fibres, carrying bits of information encoded in them. Similarly, researchers figured out that quantum information can be encoded in a photon, the “elementary particle” of light, which can be transmitted very conveniently in the same way as classical pulses of light. Photons, compared to most platform used for stationary qubits, interact very little with their surroundings. This means they don’t need to be kept cold or in vacuum, they are reasonably robust against decoherence (the corruption of the encoded quantum state) and travel at the fastest speed possible. However, their strengths are linked to their drawbacks: a very weak interaction means that it’s hard to manipulate them and perform operations to control their quantum state, and because of the intrinsic nature of light they cannot be stopped [3].
However, it is not very handy to have a great platform for a stationary qubit and amazing carriers of quantum information if they cannot talk to each other. It is a bit like having a working computer and a functional internet, but missing the cable to plug them together. We can use the photon to transmit the quantum information stored in the stationary qubit, but in order to do that, we first need to put them in an entangled state: we need to make them interact.
The problem of enhancing and controlling light-matter interaction at the single-photon and single-atom level is an old one, and definitely not an easy one. But there are recipes to make this work, so let’s take a look at what we need to make it happen.
Ingredients:
1. An optical transition – The colour centre in diamond
Photons, and light in general, have a well-defined energy. This energy defines its colour: when you see an object red, blue or green that’s nothing but its atoms absorbing and emitting photons at a very specific energy that determines their colour. This is the fundamental mechanism that allows light to interact with matter; an atom can absorb a photon’s energy, which is used to excite one of its electrons into a more energetic state, and release it again in the form of another photon that is emitted and travels away. This is called an optical transition. It can be a very selective process, as electrons in an atom can only be in discrete states that correspond to a transition with a very specific energy. So, to excite the optical transition, the photon energy needs to match that of the electronic energy levels.
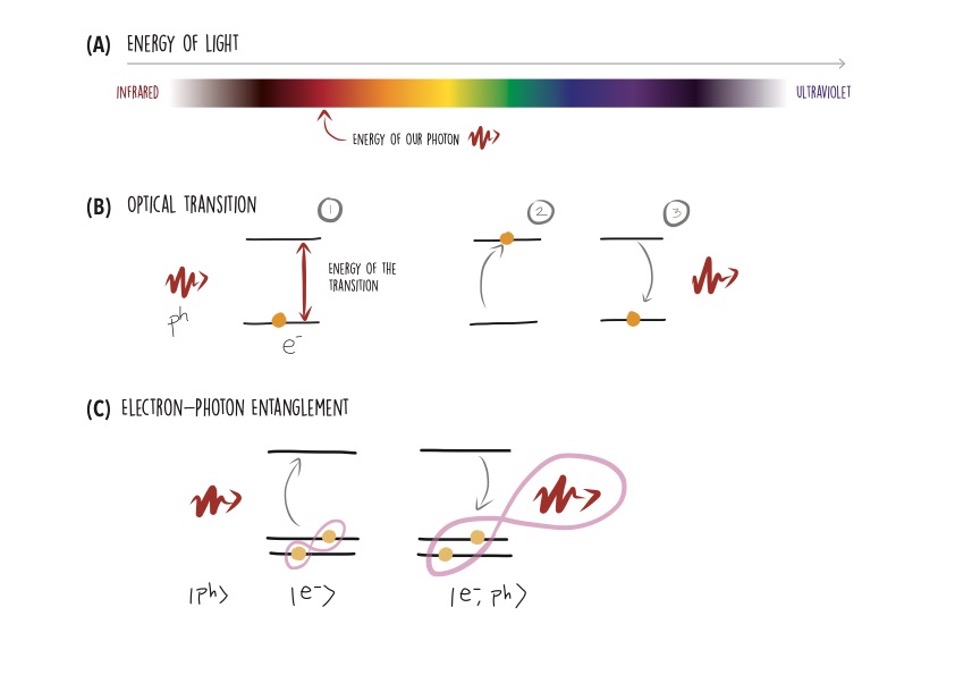
To realise stationary qubits, some platforms encode information in the quantum state of an electron. If the electron can undergo optical transitions, it is possible to connect the quantum state of the electron to the quantum state of the emitted photon.
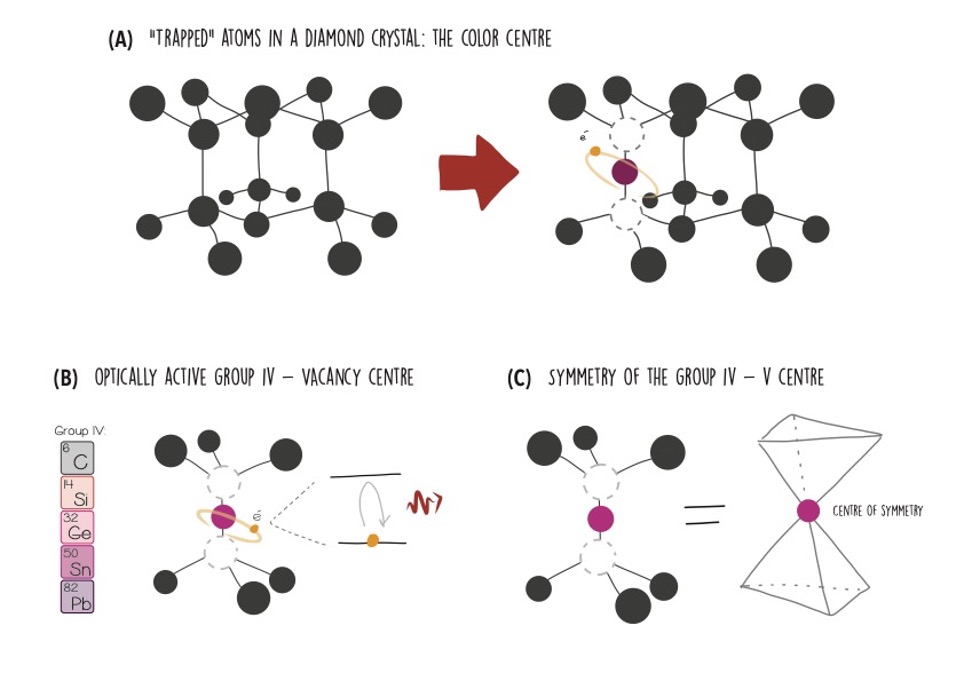
One of my favourites (not only because it is the one I work with) is the colour centre in diamond. It consists of an impurity, for example an alien atom, “trapped” inside a diamond crystal. Diamond is a very regular and ordinated crystal. Impurities that find themselves trapped inside such a crystal can behave similarly to a single isolated atom, with an electron-based qubit and, possibly, an optical transition. If you’re wondering about the name, centre is because it’s a point-like defect in a pure crystal, and colour because often the energy of its optical transition corresponds to light within the visible spectrum.
The most famous colour centre in diamond is the Nitrogen Vacancy, NV for friends, which is composed by a Nitrogen atom and a “hole” resulting from a missing Carbon atom in the diamond lattice. Other colour centres can feature different atoms.
Colour centres can be formed by processing a slab of diamond, and they can be addressed and controlled using lasers, since diamond is transparent to most of the visible light.
2. A way to trap light – Cavities
Atoms, however, and colour centres as well, are small. If a photon can travel freely in space (or inside the diamond), the probability that it will bump into our colour centre and interact with it is low. One way to overcome this problem is simply to throw many photons towards our centre, hoping that at least one of them will interact. This is realised, in experiments, by using powerful laser beams. However, this is not very efficient and it’s a probabilistic process, meaning that only one (if we’re lucky) of our photons will interact, but we don’t know for sure and we can’t tell which one.
Alternatively, we can let a single photon pass by the atom many times until eventually it will interact with it. Even though photons don’t really want to stay still, it is possible to force them to run around in a well-confined way using mirrors. By taking two facing mirrors, we can make a photon bounce back and forth in a limited space: we formed a cavity. If our colour centre is between these mirrors, the chance that at some point the photon will interact with it during one of its roundtrips is higher. We have just enhanced the interaction between light and matter.
Of course, the better the mirrors are (such that the photons will be reflected almost all the times and stay inside the cavity for longer) and the smaller the cavity volume is (and so the optical mode, that is the space where the photon is localised) the higher will the probability be of having atom-photon interactions.

2b. Bonus point: scalability – the world of photonics crystals
A smaller cavity not only increases the light-matter interaction: the simple fact that it occupies less space allows it in principle to be scalable. Why is that important? For large scale technological applications! Take the transistor as an example, key element of many electronic circuits: if it was still the size of a fist, like the first prototype developed in the fourties, electronics would not have gone very far. However, technology allowed to reduce its dimensions to only few nanometres and now we can have billions of them in the palm of our hand (the processor of your smartphone!).
Using the concept of photonics crystals [4], that exploits the behaviour of light when it reflects off a surface between two different materials and the periodic stacking of such materials, it is possible to design microscopic mirrors which are sculpted in the diamond bulk itself. This can be done by creating and repeating interfaces between diamond and air. This technique allows to carve cavities directly around the colour centres and keep their dimension as small as few hundreds of nanometres. Even better, the fabrication process allows to create thousands of such devices in a single diamond piece. The hope is that, in the future, improvements in the manufacturing processes and in the experimental techniques will allow to scale up the number of devices in the same way as it has happened for transistors in electronics.
As the size of the devices gets smaller, however, the colour centre starts to be too close to the diamond surface. This surface has a very noisy charge environment, due to electrons and defects, which can potentially disturb the electron of our colour centre causing the quality of the optical transition to degrade and the memory time of the quantum state to shorten. In the vicinity of the surface some colour centres which are very sensitive to noise in the charge environment, like the NV, lose the properties that makes them good qubits. So, if we want to use a small cavity we need…
3. A robust qubit – Introducing the Tin Vacancy
Few years ago, researchers discovered a new family of colour centres in diamond. They are made up by a substitutional impurity atom and a carbon “hole”, just like the NV, but in this case, the substitutional atom is not Nitrogen but belongs to the Group IV of the periodic table [6], that includes Silicon, Germanium, Tin and Lead. They are called “Group IV-Vacancy”, or simply SiV, GeV, SnV and PbV [7]. And unlike the NV, they form a peculiar structure that is symmetric with respect to its central point.
Often, in physics, symmetries are connected to interesting properties such as conservation laws or invariance. In this case, the central symmetry of the defect causes the Group IV – vacancy to be insensitive, to first order, to electric fields fluctuations (the linear electric dipole disappears), and thus robust against a noisy charge environment, making it a perfect fit for our small cavities integrated in diamond.
The Silicon-Vacancy (SiV) was the first one to be studied, because it is close in size to Carbon and the easiest to control and embed in a diamond crystal, but Group IV-Vacancies with heavier atoms, like the Tin-Vacancy, are expected to deliver even better performance. Indeed, one drawback of the SiV is that it is very demanding in terms of operation temperature. To make it a useful qubit, the SiV needs to stay at temperatures around 50 mK. By contrast, Tin-Vacancies (SnV) can show the same performance at around 1K, being less sensitive to heat loads in the diamond. Sounds like a small improvement, but it’s enough to make a difference in terms of experimental equipment and requirements. Making things colder gets harder and harder the more you decrease the temperature [8].
Once all the ingredients are in place, the recipe seems to work. A research group from Harvard recently witnessed an enhanced interaction between a SiV and photons inside a photonic crystal cavity In one of their latest works [9]., they sent a single photon to the cavity-SiV and managed to entangle it with the quantum state of the SiV electron, realising one of the key operations required for a node of a quantum internet.
We got to the point where we can harness quantum systems at their most fundamental level, tailoring them to suit our needs, and realising experiments where the interactions are less and less probabilistic. Researchers are learning how to efficiently interface atomic-like qubits with single photons; while this was hard to realise with colour centres in diamond until few years ago, the discovery of Group IV defects and the advances in nanofabrication techniques are paving the way for a new generation of diamond-based quantum optical devices. Maybe soon, we will get to the point where a very strong coupling between light and matter will allow deterministic spin-photon operations, where each photon sent to the system will interact with the spin. This would open up incredible possibilities both for quantum information and for fundamental research in quantum mechanics.
References:
[1] https://blog.qutech.nl/2018/09/27/a-quantum-internet-made-of-diamonds/
[2] D. P. DiVincenzo, The Physical Implementation of Quantum Computation, arXiv:quant-ph/0002077 (2000)
[3] to be more precise, it is possible to play “tricks” and slow down light while it propagates in particular materials, and effectively stop it. In practice, in this way light is “stored” into the material itself. See, for example, Ginsberg et. al., Nature. 445 (2007).
[4] E. Yablonovitch, Photonic crystals: semiconductors of light, Scientific American (2001), http://optoelectronics.eecs.berkeley.edu/eliy_SCIAM.pdf
[5] S. Mouradian et. al., Rectangular Photonic Crystal Nanobeam Cavities in Diamond, arXiv:1704.07918 (2017)
[6] Group IV indicates the Carbon group in the old notation of the periodic table, which is still commonly used in some fields of physics. The modern IUPAC name is Group 14.
[7] C. Bradac et al. Quantum nanophotonics with group IV defects in diamond. Nat Commun 10 (2019). https://doi.org/10.1038/s41467-019-13332-w
[8] https://blog.qutech.nl/2021/02/12/5-tips-for-cooler-chips/
[9] C. T. Nguyen, Quantum Network Nodes Based on Diamond Qubits with an Efficient Nanophotonic Interface, arXiv:1907.13199 (2019)
About the Author

Matteo’s interest in quantum optics brought him all the way to QuTech where, in the Hanson Lab, he spends his time playing with light and diamonds and facing the countless challenges that fill the adventurous life of an experimental PhD student.
When he’s not in the lab, surrounded by the noise of cryostats and humming equipment, he makes his own noise with a guitar or any other sound-emitting tool he finds.

