21.04.2022by David van Driel
Van Keramiek tot Quantum/From Ceramics to Quantum Mechanics 2
Van Keramiek tot Quantum/From Ceramics to Quantum Mechanics 2
How Delft became the city of quantum
by David van Driel
Bounce a coin off the walls of the Applied Physics building at the Delft University of Technology and chances are you hit a qubit. Yet when most people think about Delft, they talk about Johannes Vermeer, DSM or Delfts Blauw earthenware. How did an industrial town in The Netherlands wrestle its way to the front row of the Quantum Revolution? In this second blog post, I will trace the threads of history from the old quantum theory to Delft’s first electron beam pattern generator.
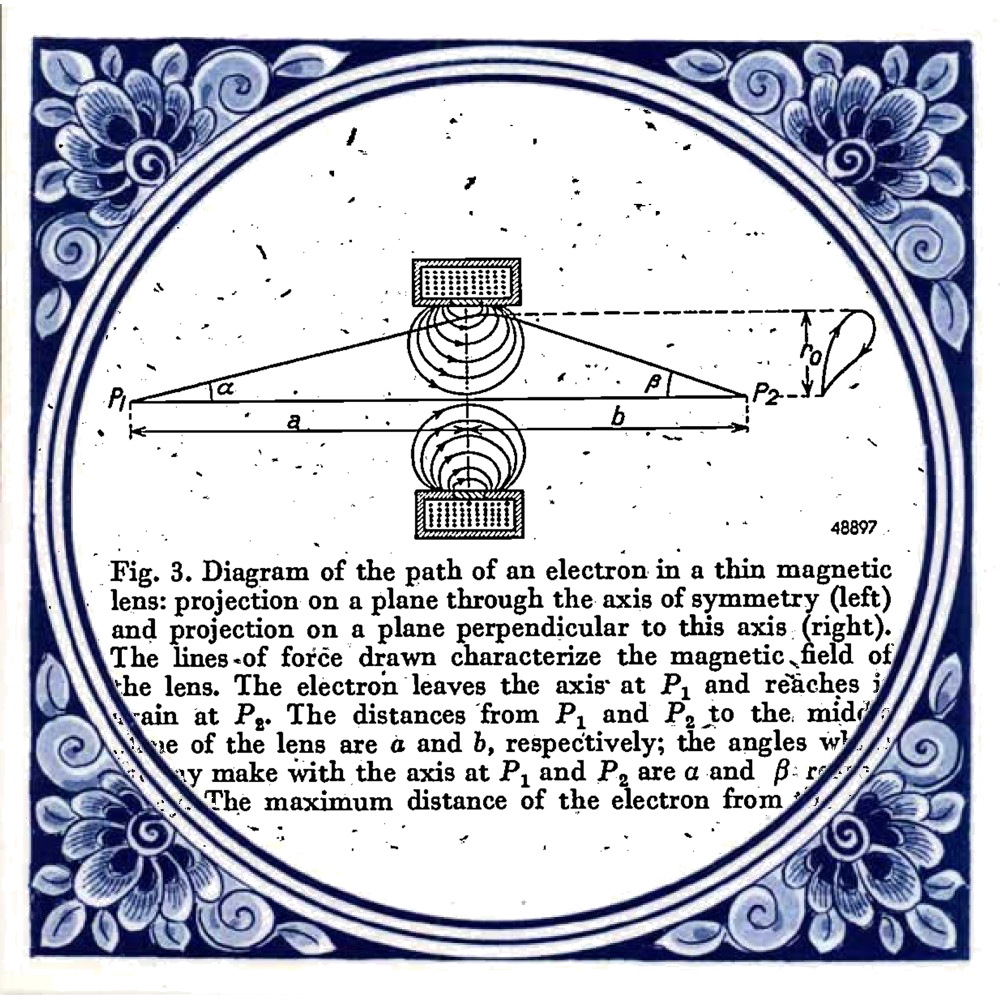
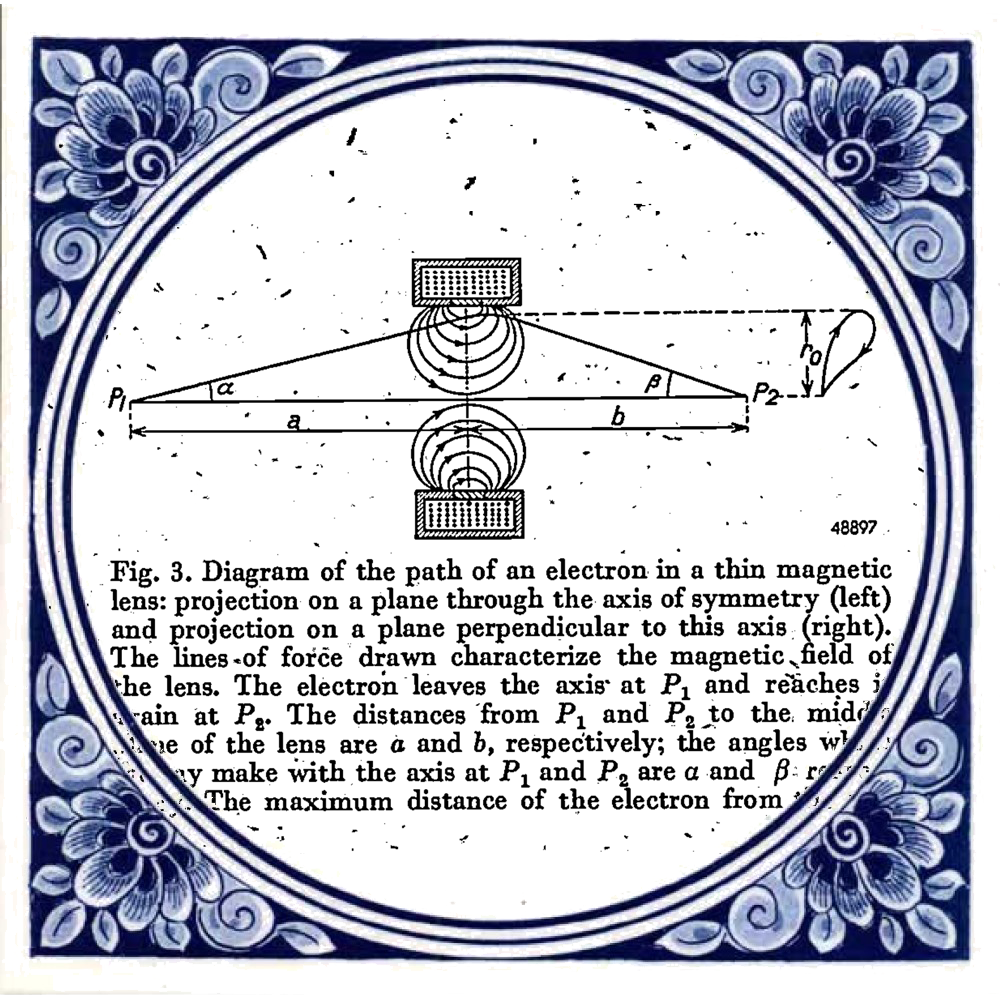
Diagram taken from: Le Poole, J. B. (1947). A new electron microscope with continuously variable magnification. Philips Tech. Rev, 9(2). Tile taken from: https://www.hollandsouvenirshop.nl/delfts-blauw-tegel-spreuk-concert-life-p-713.html
Before I start, a big thanks to Hans Mooij. He was so kind to let me interview him regarding the birth of quantum technology in Delft. I had assumptions regarding the growth of quantum technology in Delft that were simply wishful thinking. Another very big thanks to Teun Klapwijk. He gave me very valuable insights regarding the state and development of physics in Delft from the ‘70s.
Part one of this blog took the reader from the heyday of Delfts Blauw porcelain to the year 1905. In this year, Einstein published four of his most influential papers and Delft university got the right to award promotions. The Philips company had opened its doors 14 years earlier. This first decade of the 20th century was a wonderful time of discovery, with Nobel prizes ranging from radioactive decay to the X-ray. It was the finest hour of Dutch physics too; Van Der Waals, Lorentz and Zeeman shared two Nobel prizes among the three of them. In 1913, Kamerlingh Onnes would join their ranks by winning his Nobel prize for low temperature physics [1]. This was the state of physics, with Leiden University taking the cake of all the Dutch universities. To be fair, the university of Delft was only awarding doctorates for 7 years by 1913.
A remarkable commercial research institute saw the light a year after Kamerlingh Onnes’ nobel prize. The Philips Natuurkundig Laboratorium (Physics Lab) or “NatLab” was founded in Eindhoven by Gerard and Anton Philips in 1914. This was the (fundamental) research department of Philips. Note that the university of Eindhoven would only follow 42 years later. In this way, the Philips brothers[2] had a very strong impact on science and technology in The Netherlands. The first director of the NatLab was Gilles Holst [3]. He’d researched superconductivity as Kamerlingh Onnes’ assistant from 1910. He went to Eindhoven after obtaining his PhD from Pierre-Ernest Weiss in Zürich in 1914. Gilles Holst ran the NatLab until 1946, during which period he created a very strong, scientific atmosphere. The lab was modelled after AT&T’s Bell laboratories. Einstein even came to visit.

Wander de Haas in the laboratory, I have no idea what the machine behind him does.
Back to Delft, which gained her first quantum professor In 1917; Wander Johannes de Haas. His PhD advisors were Einstein and Kamerlingh Onnes. De Haas’ name lives forth in three effects; Shubnikov-De Haas, De Haas-Van Alphen and Einstein-De Haas. The first one states that the electrical resistance of metals increases upon applying a magnetic field [4]. Walk around the halls of the applied sciences building in Delft and you will still see posters showing this effect [5]. De Haas-Van Alphen states that the magnetic susceptibility of a metal oscillates as a function of magnetic field. Susceptibility is a measure of how easily one can make a metal magnetic. Paperclips are susceptible, knives aren’t. The final effect is called ‘Einstein-De Haas’. It says that a change in direction of an object’s magnetic field causes it to rotate. Although De Haas was made professor in Delft, he did his research in Leiden. This happened multiple times throughout history. De Haas would leave Delft, head to Groningen and settle in Leiden again.
In 1924, first Natlab director Gilles Holst hired a man named Henk Dorgelo to work on radio physics. Henk had just defended his PhD on gas discharge physics, a subject that would certainly benefit Philips [6]. After only 3 years, Dorgelo already became professor in Delft. His mission: the establishment of applied physics at the Technische Hogeschool Delft [7]. This field was gradually accumulating great talents, like Adriaan Fokker who was named Professor of theoretical and applied physics in 1923 [8]. Adriaan was the brother of famous aircraft engineer Anthony Fokker, also known as ‘The flying Dutchman’. Adriaan Fokker had completed his PhD under Lorentz in Leiden. The title was ‘Over Brown’sche bewegingen in het stralingsveld, en waarschijnlijkheids-beschouwingen in de stralingstheorie’. In English: “On Brownian motion in the radiation field and probabilities in radiation theory”. Herein lies the origin of the Fokker-Planck equation. This is a stochastic partial differential equation that calculates the time evolution of a particle undergoing Brownian motion. The Fokker-Einstein relation equates a particle’s diffusion constant to its movement [9]. As with De Haas, Fokker would teach in Delft but research in Leiden. He would do so until 1927, when he became assistant to Lorentz at the Teyler’s museum in Haarlem. Famous professors like Fokker and De Haas (later also Kramers and Kronig) taught physics in Delft, but quantum wasn’t in the hearts and minds of the people yet. To illustrate, a man named J.J. Koch defended his PhD titled “Applications of eigenfunctions to problems of applied mechanics” in 1929 [10]. De Broglie had received the Nobel prize for the wave nature of electrons in the same year [11]. Yet people were applying eigenfunctions not to quantum mechanics, but to applied mechanics. The Technische Hogeschool Delft was still very much an engineering college.
In 1935 a man named Jan Berend Westerdijk went to study at ETH Zürich, just like Gilles Holst had done. Westerdijk got a taste of the old quantum mechanics because he studied directly with Wolfgang Pauli. We won’t see Westerdijk in Delft for a bit, but it pays to follow his path. Meanwhile, the old quantum theory was tunneling into the heads of professors in Delft. One of receivers was E.C. Wiersma, who was appointed in 1936. He completed his PhD under De Haas. He worked on adiabatic demagnetization, low-temperature magnetism and reached a temperature of 4.4 mK [12]. Hendrik Casimir [13] described him as a ‘versatile physicist’, as you will see in the next decade. Hendrik Kramers also became professor in Delft in 1931, which he would remain until his death. Kramers is quite famous in the quantum transport community for his ‘Kramers pairs’. Imagine a bowling ball rolling along a wooden floor. Now reverse the arrow of time on the bowling ball, it will come running back to your hands. If the time-reversed bowling ball has the same energy as the ordinary one, the two form a Kramers’ pair. Hendrik Kramers was also famous for his Kramers-Kronig relations. These are mathematical relations between the real and imaginary parts of a complex function. The other name-giver of the relations was Ralph Kronig. He became professor in 1931 in Delft and was actually one of the first physicists to propose the concept of electron spin [14]. Unfortunately, Pauli, Niels Bohr and Kramers dismissed his idea and he didn’t publish on it. Think about it, would you publish an idea if those three physicists called it silly? Kronig forged a number of important theories, including the Kronig-Penney model. This is an intuitive model that can give a band gap in the dispersion relation of 1D crystals [15]. One might say that Kronig’s solid state physics in the 1930’s easily fits with the research that’s currently being done in Delft. At the time, Delft had both brilliant theoretical quantum physicists and talented electrical/physics engineers. Is this the start of the development that led us to nanotechnology? I must disappoint the reader, it’s impossible to draw one line from Bohr’s contemporaries in Delft to quantum technology nowadays. All the quantum research was being done in Leiden. “But Kramers, Kronig and De Haas taught courses in Delft right?” Yes, this is true, but Lorentz also taught physics to medical students. He certainly wouldn’t teach them second quantisation. The same goes for physics in Delft. In fact, Kronig has said that quantum mechanics wasn’t necessary for physical engineers. Hence, he refused to teach it in Delft [16].
Before we go to the 1940s, there is a very important organisation that saw the light in the 1930’s. This is the Netherlands Organisation for Applied Scientific Research, better known as ‘TNO’. The history of its foundation is too long to summarise here, but its raison d’être is clear. In the early 20th century, physics professors were called upon by the government to solve issues. An example is the research Lorentz had to do on the placement of lightning rods on the current Rijksmuseum in Amsterdam [17]. It might be a bit overkill to hire a Nobel prize winning theorist to solve such a practical problem. Yet Lorentz was also hired to work on the damming of the Zuiderzee [18].
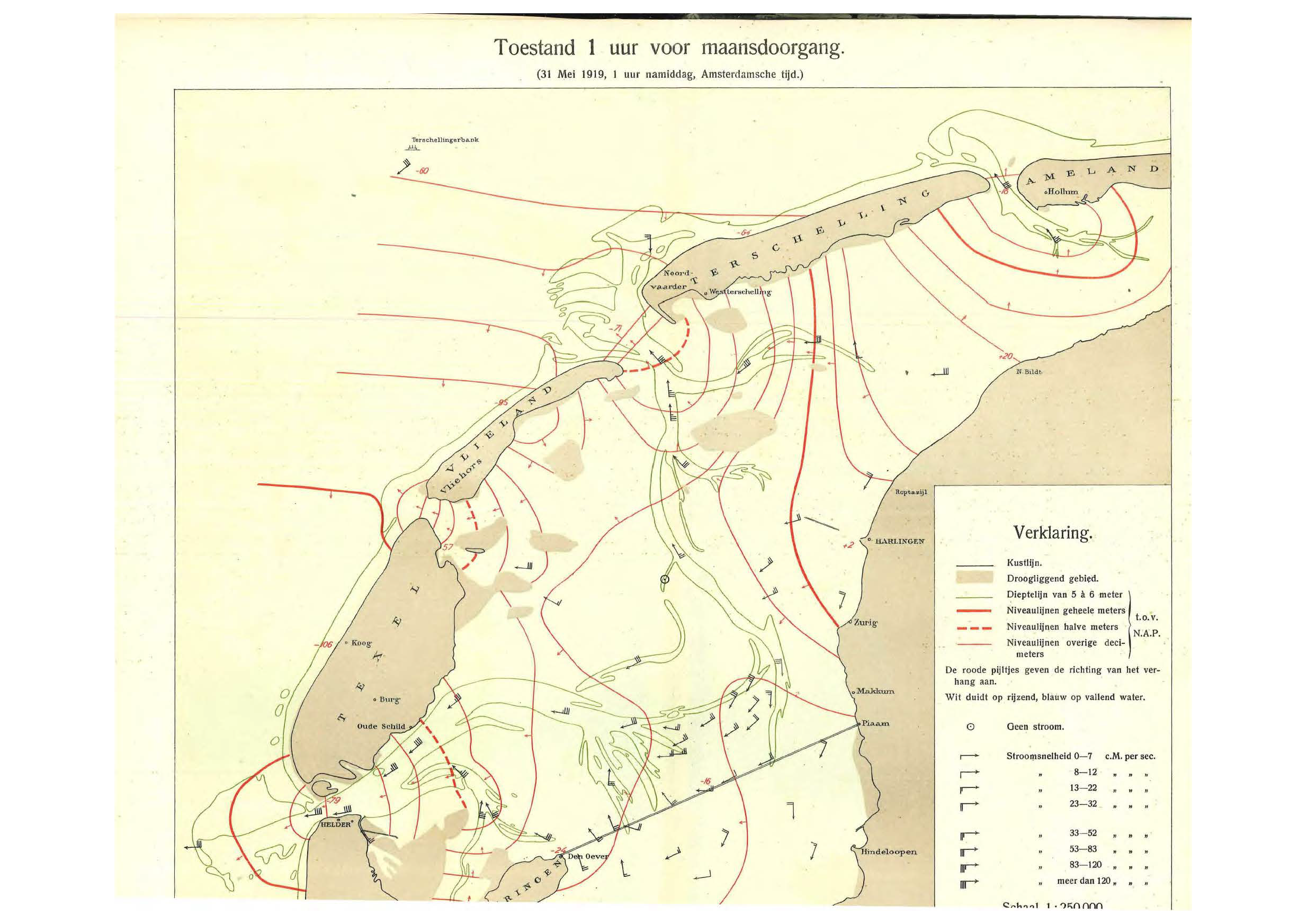
Lorentz’ calculation of the tides as a result of the damming of the Zuiderzee. The dam is the black line that connects Piaam to Wieringen. Lorentz found that this design would make the tides much more extreme, so he proposed moving the dam to Zurig instead. The fact that we dammed a sea led to the saying: “God created the world, but the Dutch created The Netherlands.”
Einstein later said that the lead engineer who got Lorentz on board had set back physics as a whole by keeping him from his theory work [19]. Regardless, it was evident that there was a dire need for applied scientific research in The Netherlands. So TNO was established in 1932. Jonathan Scheeres writes in an essay that TNO didn’t fit very well in The Netherlands at the time [20]. Its purpose would become more evident during and after the second world war.

“When the bombs drop… Whistle, Doppler-effect and impact probability”. Wartime research by Henk Dorgelo on the sounds of dropping bombs.
Professor Wander de Haas had great insight in physics, but also geopolitics. He convinced the Dutch government to purchase a large amount of uranium in 1939. During the Second World War it was hidden from the Germans in a basement of the TU Delft [21]. This was pretty brilliant, considering the German ‘Uranverein’ effort to develop nuclear weapons started during the same year [22]. A year later, The Netherlands was already in occupation by the Nazi’s until its liberation in 1945. The occupation of The Netherlands continues to have a complicated psychological and sociological effect on Dutch culture. There is even a Wikipedia page on the concepts of right (goed) and wrong (fout) in the Second World War. As such, the “goed-fout boundary” ran straight through physics and physicists. Peter Debye, known for his temperature [23], was stripped of academic honours after some of his wartime letters were rediscovered in 2006 [24]. Since the main objective of this blog is to speculate on how an old ceramics city turned into a quantum city, I will try to describe some remarkable scientific developments that took place in Delft during the War as objectively as possible, hoping not to tread the sensitive line of wrong and right.
In 1939, half of all 1700 students of the Technische Hogeschool Delft (TH Delft) were drafted into the army. There was even a ‘TH company’ military unit [25]. Two days after the Dutch surrender in May 1940, all of the students were imprisoned in Rotterdam. One day later, they are released and expected to resume studies again. Meanwhile, the TH Delft has to continue operation like before German occupation. Remember Henk Dorgelo? He’d been a professor at the TH Delft since 1927 after his research at the NatLab. In 1941, Dorgelo established the technical physical service of TNO and became its first president. This service performed research for the Dutch industry. The Nazi’s didn’t understand what TNO was due to its unique legal and independent status. Hence, it could do its work relatively unhampered [26]. A lot of research could go on thanks to TNO. Somehow, the Second World War elevated the importance of TNO, whereas its role before was less clear. Dorgelo had time to research besides his duties at TNO. In 1941, he wrote a newspaper article about using the Doppler effect to determine whether a falling bomb will hit you. An especially amusing quote is “The infrantry is right when he says that a bullet that whistles will not hit you. But bombs, thrown out of airplanes, are different.” Dorgelo became rector for the first time in 1942, this time at the TH Delft.
Sometime during the year 1940, Jan Berend Westerdijk graduated as ‘Diplom-Ingenieur’ (dipl. ing.) from Zürich [27]. He worked as assistant for the TH Delft soon afterwards. From 1942, E.C. Wiersma becomes director of the Kamerlingh Onnes laboratory in Leiden. He put all his efforts into keeping this lab in operation throughout the war. This served to keep the oppressor from stealing his scientific equipment and instrument maker students for factory work in Germany. In 1943, Westerdijk became conservator [28] of the Kamerlingh Onnes laboratory. He published with E.C. Wiersma on magnetooptics of organic substances [29].
In March 1943: all university students had to sign the ‘loyaliteitsverklaring’ (declaration of loyalty). If you sign, you declare loyalty to the Nazi’s and are allowed to continue your studies. If you don’t you are sent to Germany to work in weapon factories [30]. The senate of the TH Delft advises students to sign the loyaliteitsverklaring. E.C. Wiersma was one of the main advocates. This was interesting, considering he was extremely active in Dutch resistance and would be arrested twice throughout the war. This was life in and around the TH Delft during the second world war. Regardless and because of the occupation, research continued. There were even some significant publications. Finally, there are two exceptional master students of the TH Delft I want to highlight. They are Clemens Roothaan and Jan Bart Le Poole.
Roothaan was an electrical engineering student. He and his brother were sent to the concentration camp Vught for Dutch resistance activity [31]. Vught is a village close to Eindhoven, where Philips is situated. Some prisoners of the camp were sent to Philips for forced labor. Roothaan was allowed to do academic work for Philips because of his scientific background [32]. He was unfortunately moved to a different camp in Germany where conditions were much worse. Despite all adversity, Roothaan wrote his master thesis in prison and received his degree in 1945. While this is enough of an achievement of a lifetime, Clemens didn’t stop there. He emigrated to the US after the war and obtained his PhD under Robert Mulliken. In 1966, Mulliken would receive the Nobel Prize in Chemistry for his seminal work on quantum chemistry. Roothaan himself developed a matrix-form alternative to the Hartree-Fock equations, known as the Roothaan equations.
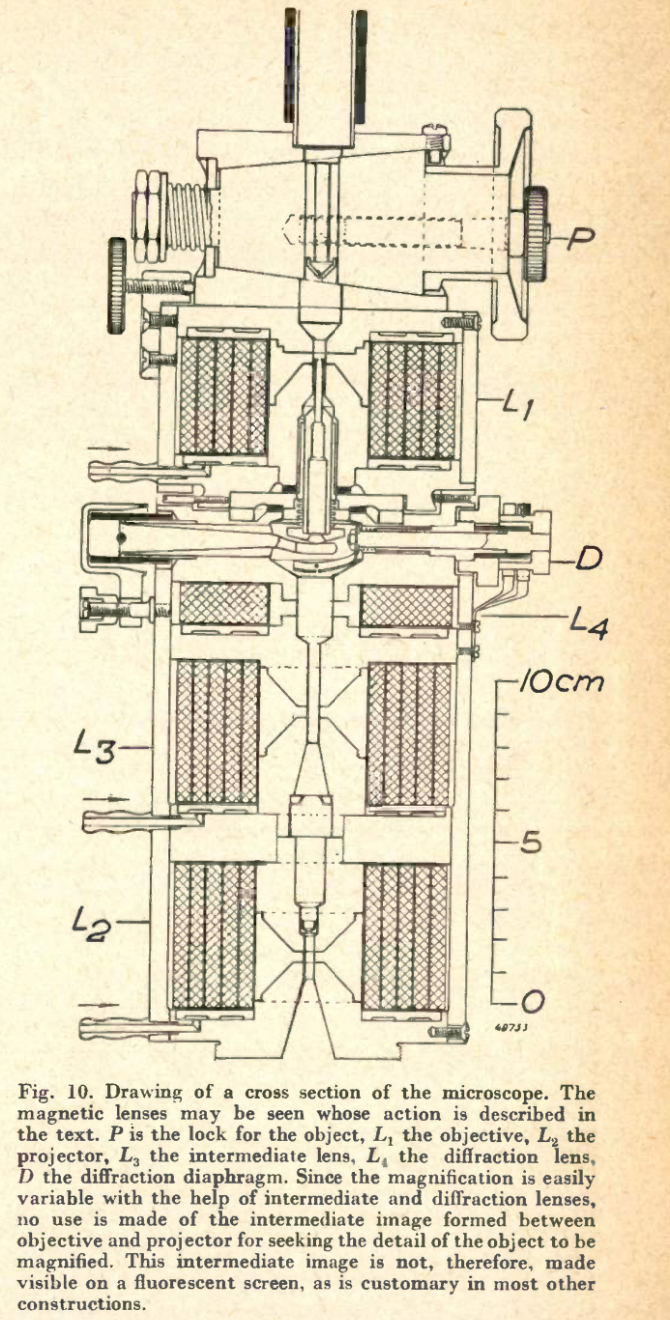
Taken from: Le Poole, J. B. (1947). A new electron microscope with continuously variable magnification. Philips Tech. Rev, 9(2).
Coming back to how the ceramics city of Delft became a quantum city, a key player in this revolution is Jan Bart Le Poole. In 1939, he started his master project with Henk Dorgelo. Although some master projects today are ambitious, this was a class of its own. He was to design and build a transmission electron microscope (TEM). Despite the invasion and occupation he made his first electron picture in 1941. He graduated the same year and continued research on a 150 kilovolt TEM. In 1944, this new prototype was completed. It was used for only a month before being taken apart to prevent the Germans from stealing it. You can see the design of the microscope in the image below.
There are two very interesting things to be found from this TEM publication. Firstly, it was written in English. Germany, and therefore German, was leading in physics publications for most of the early 20th century. The events leading up to (and of) the Second World War changed this. Secondly, note the journal “Philips Tech. Rev”. The company Philips did so much research that it decided to create its own journal. It was published yearly. This TEM publication was taken from the 1947 edition which numbered 353 pages.
Le Poole pleaded with Philips to commercialise his prototype. While initially rejected, he convinced Anton Philips himself to take it into production [33]. To this end, Philips Electron Optics was established in 1947, with Le Poole as a technical consultant. This company still exists today, but you might know it as FEI. They still sell world-leading TEM’s. Here’s where the story takes an interesting twist. I will lift a corner of the great veil of quantum in Delft [34]. Due to Le Poole’s invention, a TEM was ‘gifted’ to the TH Delft. This TEM was later refitted to become Delft’s very first electron beam pattern generator (EBPG). A machine like this allows you to do lithography on the nanoscale. A technique that gave rise to computer chips as we know them. You might wonder how this affected the events leading up to Kavli, QuTech and everything. Read my next blog post and you will find out!

David van Driel’s passion for quantum computing took him all the way from Leiden to Delft. He works on semiconductor-superconductor physics in the group of Leo Kouwenhoven. While this may sound very scientific, the daily practice amounts to putting things on top of other things (on the nano scale). He likes writing history-themed posts to not compete with the intellectual bloggers that can explain their work in 800 words. In his free time, he likes playing guitar, reading 20th century books and trying to crack the stock market with machine learning.
References and footnotes
[1] Vanpaemel, G. (2021). Frits Berends en Dirk van Delft, Lorentz. Gevierd fysicus, geboren verzoener.
[2] Gerard and Anton Philips, the founders of the Dutch multinational Philips
[3] https://nl.wikipedia.org/wiki/Gilles_Holst
[4] https://www.lorentz.leidenuniv.nl/history/explosion/dehaas.html
[5] There’s also recent publications in the quantum transport community: http://pure.tudelft.nl/ws/files/53110403/PhysRevB.96.241401.pdf
[6] https://nl.wikipedia.org/wiki/Henk_Dorgelo
[7] https://www.rhetoricadispuuttau.nl/staut/jaargang-39/henk-dorgelo/
[8] http://resources.huygens.knaw.nl/bwn1880-2000/lemmata/bwn3/fokkerad
[9] https://onlinelibrary.wiley.com/doi/epdf/10.1002/andp.19143491009
[10] https://repository.tudelft.nl/islandora/object/uuid%3Abd2a2adc-802a-4ba7-a488-2a346942d149
[11] https://www.nobelprize.org/prizes/physics/1929/summary/
[12] https://www.sciencedirect.com/science/article/abs/pii/S0031891434900021
[13] Dutch physicist and discoverer of the eponymous Casimir effect.
[14] A rolling bowling ball also spins. Yet, the electron spin is a fundamental property which every electron has, regardless of their energy or velocity.
[15] de L. Kronig, R.; Penney, W. G. (3 February 1931). “Quantum Mechanics of Electrons in Crystal Lattices”. Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences. The Royal Society. 130 (814): 499–513. doi:10.1098/rspa.1931.0019. ISSN 1364-5021.
[16] From personal communication with Hans Mooij
[17] Ingekomen en minuten van uitgaande brieven gewisseld met architect Cuypers, H. Haga en anderen, betreffende bliksemafleiders aan het Rijksmuseum te Amsterdam en andere gebouwen; met theoretische aantekeningen, 1888, 1906-1907, 1911
[18] Map from: Lorentz, H. A. (1926). Verslag van de commissie Lorentz (gevolgen afsluiting Zuiderzee op het getij). scan van 10042488. For the origin of the quote, see: http://vakbladvitruvius.nl/images/essay/TheDutchMade_F.Niemeijer_May2021_DEF-dd27mei.pdf
[19] https://www.delta.tudelft.nl/article/uit-delftse-bronnen-eredoctors
[20] https://essay.utwente.nl/66556/
[21] http://resources.huygens.knaw.nl/bwn1880-2000/lemmata/bwn1/haas
[22] Monk, Ray. “Robert Oppenheimer: His life and mind (a life inside the center).” (2013): 848.
[23] Also known for his salary. Casimir said that people used ‘milli-Debye’ as a unit for salaries of average physicists.
[24] https://physicstoday.scitation.org/doi/10.1063/1.2216953
[25] For an expansive overview, read this brilliant article: https://www.delta.tudelft.nl/article/vijf-verwarrende-jaren
[26] https://www.espunt.nl/Mijn-schrijfproducten/Artikelen,-columns-e-d/Oorlogsnood-vormde-TNO/mobile/
[27] https://www.hogenda.nl/wp-content/plugins/hogenda-search/download_attachment.php?id=526&type=biography
[28] In my view, this is an independent researcher with the additional responsibility to ‘conserve’ the laboratory
[29] Broersma, S., Waterman, H. I., Westerdijk, J. B., & Wiersma, E. C. (1943). Determination of the magnetooptic rotatory power of pure organic substances and their mixtures. Physica, 10(3), 97-129.
[30] https://oorlog.arq.org/nl/nieuws/zou-jij-je-een-loyaliteitsverklaring-aan-de-nazis-tekenen
[31] https://nl.wikipedia.org/wiki/Clemens_Roothaan
[32] https://news.uchicago.edu/story/clemens-cj-roothaan-eminent-quantum-chemist-and-concentration-camp-survivor-1918-2019
[33] https://nl.wikipedia.org/wiki/Jan_Bart_Le_Poole
[34] Einstein famously said that De Broglie had “lifted a corner of the great veil”: https://history.aip.org/exhibits/einstein/quantum1.htm